-
Tuning symmetry breaking charge separation in perylene bichromophores by conformational control
A. Aster, G. Licari, F. Zinna, E. Brun, T. Kumpulainen, E. Tajkhorshid, J. Lacour and E. Vauthey
Chemical Science, 10 (2019), p10629-19639


DOI:10.1039/C9SC03913A | unige:126803 | Abstract | Article HTML | Article PDF | Supporting Info
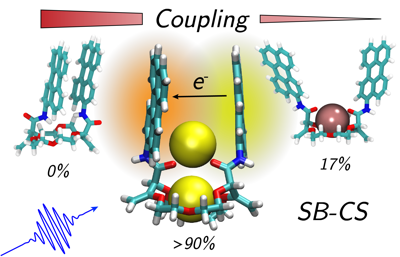
Understanding structureâââ‰â¬Åproperty relationships in multichromophoric molecular architectures is a crucial step in establishing new design principles in organic electronics as well as to fully understand how nature exploits solar energy. Here, we study the excited state dynamics of three bichromophores consisting of two perylene chromophores linked to three different crown-ether backbones, using stationary and ultrafast electronic spectroscopy combined with molecular dynamics simulations. The conformational space available to the bichromophores depends on the structure and geometry of the crown-ether and can be significantly changed upon cation binding, strongly affecting the excited-state dynamics. We show that, depending on the conformational restrictions and the local environment, the nature of the excited state varies greatly, going from an excimer to a symmetry-broken charge separated state. These results can be rationalised in terms of a structureâââ‰â¬Åproperty relationship that includes the effect of the local environment.